Lymphoma

Currently, there is no clearly defined standard of care (SOC) for patients with relapsed/refractory follicular lymphoma (R/R FL).

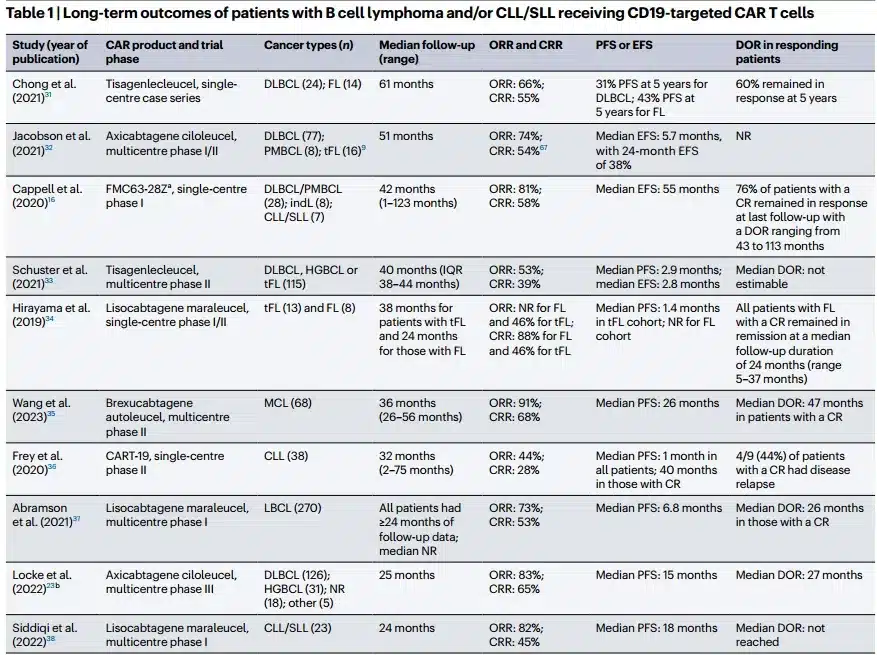
Kymriah (Tisagenlecleucel) is an autologous anti-CD19 chimeric antigen receptor (CAR) T-cell therapy that has shown clinically meaningful results in patients with relapsed/refractory B-cell lymphoma.

Promising News from Cellular Therapy. On March 29, 2024, Ms. Mo (alias), a patient with diffuse large B-cell lymphoma who received CAR-T cell therapy, was discharged from the Department of Hematology and Rheumatology at the People’s Hospital of Zhuhai.

On May 15th, 2024 (local time), the U.S. Food and Drug Administration (FDA) announced the accelerated approval of Breyanzi (lisocabtagene maraleucel), Bristol Myers Squibb’s CAR-T therapy, for an expanded indication to treat adult patients with relapsed or refractory follicular lymphoma (FL) who have received at least two prior systemic therapies.

According to a retrospective study presented at the 2024 Transplantation & Cellular Therapy Meetings, a single infusion of brexucabtagene autoleucel (brexu-cel) achieved a higher rate of central nervous system (CNS) remission in patients with relapsed/refractory B-cell acute lymphoblastic leukemia (R/R B-ALL) and active CNS involvement.

The American Association for Cancer Research (AACR) Annual Meeting is the most influential oncology scientific event globally. From population science and prevention, to cancer biology, translational medicine, and clinical research, to survivorship and advocacy, the AACR Annual Meeting showcases the outstanding achievements of cancer research institutions worldwide.

CD19 CAR-T cell therapy has significantly improved the prognosis of patients with relapsed/refractory large B-cell lymphoma (r/r LBCL), including diffuse large B-cell lymphoma (DLBCL), primary mediastinal B-cell lymphoma (PMBCL), and LBCL transformed from follicular lymphoma (t-FL) or non-follicular lymphoma (t-NFL).