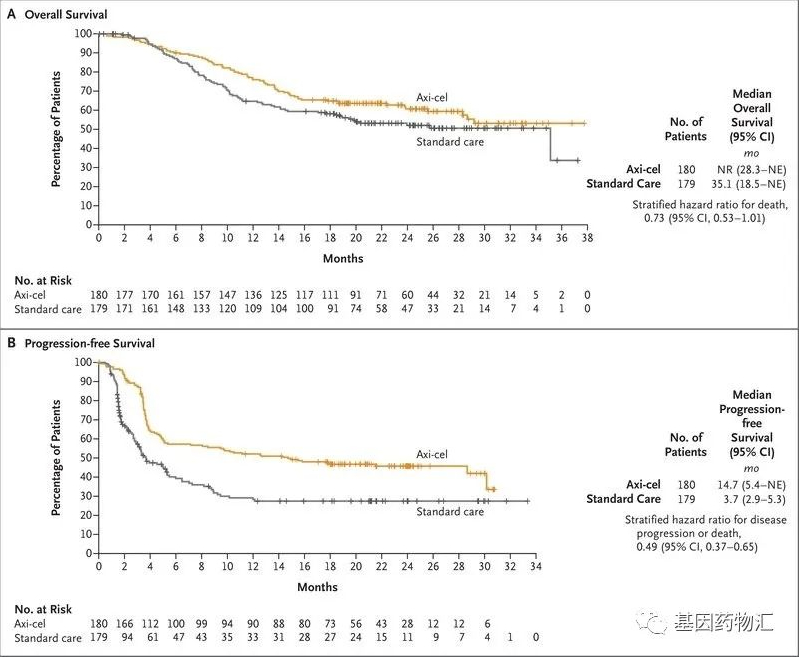
As of May 1, 2018, the FDA has approved Novartis’ CD19-targeted CAR-T therapy tisagenlecleucel (Kymriah) for the treatment of adult patients with relapsed or refractory large B-cell lymphoma after two or more lines of systemic therapy, including diffuse large B-cell lymphoma (DLBCL), high-grade B-cell lymphoma, and DLBCL arising from follicular lymphoma.