Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

🚀Revolutionizing Medicine: CAR-T Therapy Beyond Cancer 🚀
🔬 Over the past decade, CAR-T cell therapy has transformed the field of oncology, successfully treating previously incurable blood cancers. While CAR-T therapy gained fame for its success in cancer treatment, the roots of this groundbreaking principle trace back nearly 30 years—initially exploring T cell therapy for HIV/AIDS. Although the early attempts didn’t succeed in treating HIV/AIDS, they demonstrated the enduring potential of engineered T cells in immunocompromised patients.
🌟 CAR-T therapy, or Chimeric Antigen Receptor T-cell Immunotherapy, essentially involves reengineering a patient’s immune T cells outside the body to recognize antigens on the surface of tumor cells. These modified cells are then reintroduced into the patient, effectively identifying and eliminating cancer cells. Scientists have been tirelessly working to extend CAR-T therapy beyond blood cancers to various other types.
💡 Moreover, research from both clinical and preclinical studies indicates that CAR-T therapy holds great promise beyond cancer treatment. It shows potential applications in autoimmune diseases, chronic infections, heart diseases, aging-related conditions, and more.
🌐 In recent years, scientists have focused on applying CAR-T therapy to solid tumors, exploring its potential in cancer types beyond blood-related malignancies. Early research results suggest that the next frontier for CAR-T therapy may extend to various fields outside cancer, including autoimmune diseases (systemic lupus erythematosus, pemphigus vulgaris, multiple sclerosis, type 1 diabetes, asthma), fibrotic diseases (fibrosis in the heart, liver, lungs, and kidneys), aging-related conditions (liver fibrosis, solid tumors, atherosclerosis, natural aging), and infectious diseases (HIV/AIDS, hepatitis B and C, pulmonary tuberculosis).
🚀 The future of CAR-T therapy holds vast potential in reshaping the landscape of medical treatment, reaching far beyond the realms of cancer. Stay tuned for ground
#immunotherapyrevolution #CARTTherapy #MedicalInnovation #BeyondCancer #ImmunotherapyRevolution #CART #cancer #tumor #cancertreatment #tcell
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

🌟China Leading Global Advances in Breast Cancer Immunotherapy 🌟#ChinaInMedicine #BreastCancer

Breast Cancer
Since 2020, breast cancer has surpassed lung cancer, becoming the most common cancer globally with approximately 2.3 million new cases and 680,000 deaths annually. Despite improvements in traditional treatments like surgery, chemotherapy, and radiation, the mortality rate remains high. China’s Chimeric Antigen Receptor (CAR) immunotherapy is making significant strides, spearheading innovation in breast cancer treatment. 🇨🇳💪
Recent studies indicate substantial breakthroughs in CAR-T therapy, as well as CAR-NK cells and CAR macrophages for breast cancer treatment. The application of combination therapies further enhances the cytotoxicity of CAR-based cell therapies against breast cancer cells, bringing new hope to patients. 🚀
China is at the forefront of global clinical research on CAR, with 16 clinical trials focused on CAR-T or CAR-NK cell therapies currently underway. Particularly noteworthy is the research at the People’s Liberation Army General Hospital, where CAR-T cells targeting CD133 are employed to treat refractory advanced malignant tumors, including breast cancer. This approach has demonstrated excellent anti-tumor activity and manageable safety in patients previously treated for advanced hepatocellular carcinoma and cholangiocarcinoma. 🏥✨
On another front, the clinical study at Sun Yat-sen Memorial Hospital, Zhongshan University, is equally intriguing. They are exploring PD-1 knockout CAR-T cells targeting MUC1 for treating late-stage breast cancer patients. This trial is a dose-escalation exploratory study, and researchers anticipate its results to pave the way for future treatments. 🧪🔍
China is not only a trailblazer in breast cancer immunotherapy research but also a miracle creator, bringing new hope to cancer patients worldwide. Let’s witness China’s outstanding achievements in the medical field and acknowledge its contribution to global health! 💊🌏
#ImmunotherapyAdvancements #BreastCancerResearch
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

🌈🌈Lyn’s Triumph Over Lymphoma: A CAR-T Journey in Shanghai! 🌟🌟 #CARTVictory

⚡️⚡️Lyn, a mother of three from Singapore, faced unexpected health challenges in 2022. She experienced persistent pain in her shoulders, neck, and upper limbs, disrupting her sleep and even affecting her ability to breathe. A visit to the emergency department at Shanghai Jiahui International Hospital revealed a shocking discovery – a massive tumor in her mediastinum, measuring 15 cm in diameter! The compression from this large mass not only caused excruciating pain but also impacted nearby organs such as the heart, major blood vessels, and lungs, leading to Superior Vena Cava Syndrome (SVCS). Lyn struggled with breathing difficulties, posing an imminent threat to her life.
🌸🌸The diagnosis pointed to “Primary Mediastinal Large B-cell Lymphoma,” a relatively uncommon subtype of lymphoma, prevalent among young individuals, particularly females. Despite undergoing various comprehensive treatments, including chemotherapy combined with targeted therapy and consolidative radiotherapy, Lyn unfortunately showed no response to these treatments. Eventually, the decision was made to pursue CAR-T cell therapy.
🧶🧶Lyn’s husband emphasized, “The cost of CAR-T treatment abroad is significantly higher than in China, and the waiting period is at least 3-6 months. We trust the medical expertise of China and Shanghai Jiahui. Their professionalism and meticulous care left a deep impression on us. Our decision was absolutely correct.”
💥💥 The treatment team at Shanghai Jiahui International Hospital, led by Director Vicky H. Lee and Dr. Lili Zhou, the head of the Hematology and Oncology Department, developed a meticulous treatment plan to navigate Lyn through the entire CAR-T therapy. They highlighted the critical importance of two stages: the period from cell collection to pre-infusion and the phase post-infusion, which poses a risk of Cytokine Release Syndrome (CRS) and Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS).
🍁🍁 After careful preliminary treatments, infection screening, and confirmation through cell collection, Lyn received the CAR-T cell infusion. Following infusion, the “T-cell soldiers” proliferated successfully in her body, providing relief from tumor-related pain. Upon clinical assessment confirming stable conditions, Lyn smoothly transitioned to being discharged.

⛵️⛵️ It’s worth noting that CAR-T therapy costs comprise drug and non-drug expenses, with the overall treatment expenses significantly lower in China compared to Western countries. Shanghai Jiahui International Hospital collaborates with local high-quality medical resources such as Ruijin Hospital and Tongji Hospital, offering patients a comprehensive one-stop CAR-T diagnostic and therapeutic service.
#CART #SurvivorStories #MedicalMiracle #Inspiration #JiahuiInternationalHospital #CancerSurvivor #CARTTherapy #HealthJourney #advancedmedicineinchina #chinesemedicine
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

🥰Chinese CAR-T Therapy: A Beacon of Hope for Lymphoma Patients🥰
🌞In a groundbreaking development, the Hematology Department at Peking Union Medical College Hospital has achieved remarkable treatment outcomes with commercial CAR-T therapy since 2021.
🌞Meet Mr. Li, a 59-year-old with a history of hypertension. In September 2019, he noticed swelling in his right elbow, which worsened over three months, prompting concerns. After visits to multiple hospitals, he was diagnosed with Diffuse Large B-Cell Lymphoma (DLBCL). Further consultations revealed additional challenges: the presence of TP53 and MYD88 gene mutations, adding complexity to his case.
🌞Local hospitals initiated frontline treatments, utilizing drugs such as cyclophosphamide, prednisone, ibrutinib, and rituximab. While the initial treatment provided relief, it was short-lived. After six months of rest, Mr. Li experienced discomfort in his lower abdomen, leading to a subsequent diagnosis of DLBCL metastasis. Despite second, third, and fourth-line treatments at local hospitals, his condition remained uncontrolled.
🌞In December 2021, Mr. Li placed his last hope in the hands of the lymphoma team at Peking Union Medical College Hospital and underwent CAR-T therapy.
On December 27, 2021, the patient underwent a successful single-cell collection, followed by CAR-T cell infusion on January 20, 2022. Post-infusion, all indicators returned to normal, with only transient fever and CAR-T-related neutropenia observed. Targeted treatment promptly addressed these symptoms, leading to their resolution. One month after discharge, a complete response (CR) was achieved upon evaluation.
🌞Professor Wang Wei, drawing on clinical expertise, emphasized that:
TP53-mutated patients often exhibit limited response to conventional frontline treatments, making CAR-T therapy a potential benefit.
🌞For high-risk patients, especially those with dual expression and recurrence within 12 months, CAR-T therapy as a second-line treatment may yield benefits.
Post-CAR-T hematopoietic suppression, while common, requires nuanced management based on various factors. R-CHOP shows safety and efficacy in R/R DLBCL. Post-CAR-T bone marrow suppression is influenced by factors like prior chemotherapy lines, occurrence of CRS/ICANs, necessitating comprehensive clinical assessment and supportive care.
🌞In summary, CAR-T therapy presents a promising avenue for patients facing challenging lymphoma scenarios. The successful case of Mr. Li underscores the potential efficacy and safety of this innovative treatment at Peking Union Medical College Hospital.

#CART #Success #LymphomaTreatment #MedicalAdvancements #pekingunion #Chinesecart
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79
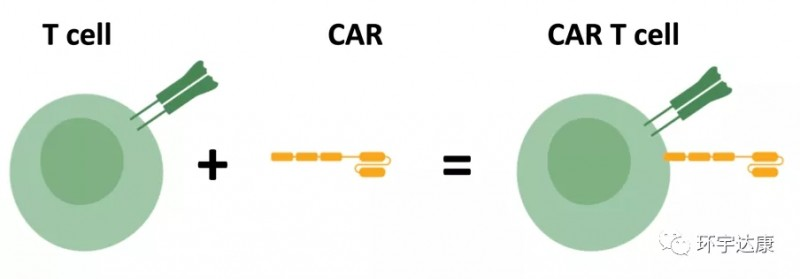
Beacon of Hope in Cancer Treatment
😊😊😊 What is the treatment process for CAR-T cell therapy?
CAR-T cell therapy has emerged as a global miracle in cancer treatment, capturing widespread attention. By loading CAR onto T cells, these “super soldiers” can specifically identify and efficiently eliminate tumor cells, offering a fresh perspective for cancer patients.
💙Assessment: Doctors first evaluate whether the patient is suitable for CAR-T therapy, ensuring maximum benefit.
❤️Isolation and Sampling: T cells are extracted from peripheral blood, preparing for subsequent modifications.
💛Cell Modification: T cells are activated and modified in vitro, using biotechnological techniques to load the CAR structure for specific tumor cell recognition.
🧡Ex Vivo Expansion: Depending on the patient’s weight and treatment period, CAR-T cells are significantly expanded ex vivo to ensure an adequate quantity for treatment.
💚Infusion into the Body: The expanded CAR-T cells are reintroduced into the patient’s body, allowing the “super soldiers” to combat tumors.
💜Subsequent Monitoring: Close monitoring of the treatment’s effectiveness post CAR-T cell infusion, promptly addressing any adverse reactions to ensure optimal patient outcomes.
The advent of CAR-T cell therapy brings a new dawn for cancer patients, with its unique treatment mechanism and remarkable effects positioning it as a leader in the field of cancer immunotherapy. This scientific marvel is spreading hope globally, opening a gateway to recovery for cancer patients.
#CARTCellTherapy #CancerTreatment #HopeInScience #CART #Cancer #Whatiscancer #cancerpatient
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

A Miraculous Journey: Israeli Artist Finds Cure for Multiple Myeloma in Hangzhou China
In a remarkable medical story, we turn our attention to the experiences of a friend seeking treatment in Hangzhou China. Tali, a well-known artist from Israel. In 2012, she was diagnosed with multiple myeloma, a malignant blood disease, and after seeking treatment in various European countries, including Israel and France, she found no definitive cure. In a moment of despair, she discovered a solution from Hangzhou, China, through the European Bone Marrow Transplant Association.

On October 6th, Tali and her family boarded a flight from Israel to Hangzhou. Under the meticulous care of Professor Huang He and his team at Zhejiang University’s First Hospital, Tali underwent over a month of intensive treatment. Today, Tali is finally on the road to recovery, expressing her gratitude to the medical staff through her preferred medium – art.

In the hospital room, Tali and Professor Huang He share laughs and conversations. The weather looks promising, initially being skeptical about her health when arriving in Hangzhou, encouragement from the medical team reignited her hope for recovery. Using her artistic language, she documented every moment of the treatment process.
After undergoing treatment, the excruciating bone pain gradually lessened, indicating a positive turn in her health.

“I can feel my body improving bit by bit. Green cells are gradually replacing the red ones, and my bone pain has completely disappeared. There are no tumor cells in the bone marrow anymore – they have vanished entirely.” Tali expressed.

The advanced treatment utilized at Zhejiang University’s First Hospital involves cutting-edge blood cell separation technology. Lymphocytes are extracted and genetically engineered to attack malignant tumor cells, successfully curing multiple myeloma. This revolutionary technique is known as CAR-T cell therapy. Zhejiang University’s First Hospital stands out as one of the earliest and most experienced clinical research centers using this technology, making it a pioneer in the field.

In here we bring you this inspiring story from Hangzhou, where art, science, and the human spirit come together in a tale of triumph over adversity.

Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79
Breakthrough CAR-T Cell Therapy for R/R B-Cell ALL: A Game-Changer in Chinese Medical Innovation
ALL- Acute Lymphoblastic Leukemia
The Fourth China Hematology Development Conference – CAR-T Frontier Forum and the First CAR-T Cell Immunotherapy Summit (JCIS), held on January 5, 2024, in Tianjin.
Potential Best-in-Class: Inaticabtagene Autoleucel Redefining Long-term Outcomes for R/R B-ALL
Professor Ma Jun from the Harbin Institute of Hematology and Oncology shared notable progress in immunotherapy and cell treatment for Chinese ALL. Previously, the overall complete response (CR) rate for adult R/R ALL treatment in China was approximately 40%, with a mere 11% 3-year survival rate. The introduction of CAR-T cell therapy has been a paradigm shift, altering the long-term outcomes for R/R B-ALL patients.
Inaticabtagene Autoleucel demonstrates superior efficacy:
Higher overall response rates (ORR) at 3 months and beyond, with median duration of response (DOR) and overall survival (OS) surpassing other products.
Patients treated with Inaticabtagene Autoleucel exhibit similar long-term benefits in OS, whether or not they undergo subsequent transplantation.
Inaticabtagene Autoleucel boasts enhanced safety:
Lower incidence rates of grade 3 cytokine release syndrome (CRS), grade 3 immune effector cell-associated neurotoxicity syndrome (ICANS), and infusion-related mortality compared to other CAR-T products.
Moreover, in minimal residual disease-positive (MRD+) B-ALL patients, CAR-T cell therapy has shown significant progress. It eradicates MRD, improves survival rates, and may serve as a first-line consolidation therapy for CR patients, aiding in:
Reduced transplant requirements, mitigating transplant-related complications
Maintenance of long-term remission for those unsuitable for allo-HSCT or unwilling to undergo it
Improved overall survival
Lower intensity and duration of intensive chemotherapy, leading to shorter treatment times and enhanced compliance.
The Future Outlook: Believing in the potential of CAR-T cell therapy, it is anticipated that this innovative treatment will extend hope to currently incurable diseases such as solid tumors and brain tumors. The strides made in Chinese medical innovation, exemplified by Inaticabtagene Autoleucel, signal a promising future for the global landscape of CAR-T cell therapy.
#CARTRevolution #InaticabtageneAutoleucel #HematologicCancerTherapy #ClinicalBreakthrough #ChinaHematologyConference #JCIS #BAllTreatment #MedicalAdvancements #CancerResearch #TreatmentInnovation #InaticabtageneAutoleucel #Autoleucel #ChineseCART #CARTTherapy #cancer #Bloodcancer #MedicineinCHINA #Medicaltourismo #Advancedmedicine #cancertherapy #leukemia
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

“Clinical Breakthrough: Chinese CAR-T – Inaticabtagene Autoleucel Revolutionizing Hematologic Cancer Therapy”
On January 5, 2024, the Fourth Chinese Hematology Development Conference was convened in Tianjin, China, concurrently hosting the CAR-T Frontier Forum and the First CAR-T Cell Immunotherapy Summit (JCIS). The focus was directed towards the latest advancements and clinical applications of Inaticabtagene Autoleucel, presenting new pathways for standardized treatments.
Under the moderation of Professors Wu Depei and Hu Yu, Professor Wang Ying from the Institute of Hematology at the Chinese Academy of Medical Sciences presented a specialized lecture titled “Interpreting Key Clinical Data of Inaticabtagene Autoleucel.” Professor Wang highlighted the challenges faced by adult B-ALL patients in China, commonly treated with salvage chemotherapy ± hematopoietic stem cell transplantation. However, the median survival period is only 2-6 months, and targeted therapies yield a median survival of merely 7.7 months, necessitating an urgent need for more effective treatment methods. Recently, the Inaticabtagene Autoleucel infusion has been approved for treating adult R/R B-ALL patients.
Critical clinical research (NCT04684147) has revealed the substantial outcomes achieved with a single treatment of Inaticabtagene Autoleucel:
Rapid and profound remission: Within 3 months of treatment, the overall response rate (ORR) reached an impressive 82.1%, with a 100% negativity rate for minimal residual disease (MRD), showcasing the remarkable effects of Inaticabtagene Autoleucel within a short span.
Enduring remission: The 3-month post-treatment overall remission rate stood at 64.1%, with a 12-month sustained remission rate (DOR) of 80%. With a median follow-up of 8.0 months, the median relapse-free survival (RFS) period has not been reached. The one-year survival rates for overall infused patients, those achieving complete response (CR)/complete response with incomplete hematological recovery (CRi) within 3 months, and those reaching CR/CRi at 3 months were 67.9%, 72.0%, and 85.6%, respectively.
Good safety profile: The incidence rate of ≥3-grade cytokine release syndrome (CRS) was only 10.3%, and that of ≥3-grade immune effector cell-associated neurotoxicity syndrome (ICANS) was merely 7.7%. Moreover, patients recovered post-treatment without complications.
Furthermore, data from over 100 patients validated the clinical efficacy and safety of Inaticabtagene Autoleucel, affirming its robustness and offering renewed hope to a larger patient population. This breakthrough treatment provides a new avenue for tackling challenging diseases and is poised to revolutionize the landscape of hematologic cancer therapy.
#CARTRevolution #InaticabtageneAutoleucel #HematologicCancerTherapy #ClinicalBreakthrough #ChinaHematologyConference #JCIS #BAllTreatment #MedicalAdvancements #CancerResearch #TreatmentInnovation #InaticabtageneAutoleucel #Autoleucel #ChineseCART #CARTTherapy #cancer #Bloodcancer #MedicineinCHINA #Medicaltourismo #Advancedmedicine #cancertherapy
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

Exploring Tumor Vitality: Chinese CAR-T Therapy Grants Patients Complete Remission
“Is it true? It’s so unbelievable!” said Chen, the “fortunate one.”
On June 22, 2021, the approval of China’s first CAR-T cell therapy product, Yescarta, marked a significant milestone in medicine. Behind this milestone lies the story of a patient named Chen, diagnosed with diffuse large B-cell lymphoma, which not only brought confidence to patients but also injected fresh belief into medical professionals.
Chen was diagnosed with diffuse large B-cell lymphoma in July 2019, underwent primary treatment, only to unfortunately relapse. Due to the insufficient efficacy of secondary treatment and considering the TP53 mutation, conventional treatment plans were inadequate. However, the approval of Yescarta on June 22, 2021, brought a glimmer of hope for patients. Under the meticulous planning of the expert team in the Hematology Department of Ruijin Hospital, Chen successfully underwent CAR-T cell infusion therapy on August 2.
One year after the evaluation of the treatment’s effectiveness, Chen’s condition still maintains complete remission. This achievement has brought immense hope and joy to patients with diffuse large B-cell lymphoma, strengthening the belief in the potential of CAR-T therapy among medical practitioners.
“This is not only good news for patients but also an encouragement and boost to us, clinical doctors in the field of hematology,” stated Professor Xu Pengpeng.
This case represents not only a medical breakthrough but also serves as an inspiration for individuals battling diffuse large B-cell lymphoma. The success of CAR-T therapy reveals new possibilities, offering a new pathway to break the limitations of the “mere six-month survival period” in cancer treatment. May more patients benefit from this breakthrough, offering hope for healing and long-term remission.
#CARTtherapy #TumorVitality #CancerTreatment #NewMedicalTechnologies #MedicalAdvancement #Bloodcancer #cancersuvivor #lymphoma
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

“Two Years of Complete Remission, CAR-T therapy has given her a new lease on life. She Thought the Journey Was Over”

Yun is a 78-year-old patient who achieved a continuous complete remission for two and a half years despite relapsed/refractory diffuse large B-cell lymphoma. Diagnosed in 2019, after enduring six rounds of chemotherapy, she initially achieved complete remission as confirmed by her healthcare team. Unfortunately, the period of remission was short-lived as Yun soon experienced disease relapse, and subsequent second-line treatments failed to yield positive results.
“I vividly remember when Director Wang Li encouraged me, mentioning an advanced method called Car-T, but it was quite expensive. I was hesitant, but I discussed it with my son when I got home. When my son and daughter-in-law heard about it, they insisted on treatment. My son said, ‘Mom, you’re the only mother I have in this world. As long as you’re here, our home is complete. Money can be earned again, and if the treatment isn’t successful, at least we won’t have regrets.’ I was deeply moved. At my age, I’ve already shown strength through previous treatments. I believe that wherever my health takes me is where I belong. Everyone’s support gave me a reason to stay strong again.”
In 2021, CAR-T cell therapy was approved and launched in China, and Yun underwent this treatment at Ruijin Hospital.
Dr. Wang Li, Director of Hematology at Ruijin Hospital, explained, “CAR-T cell therapy provides new treatment options for a wide range of cancer patients.”
“Before planning Yun’s CAR-T cell therapy, lymphoma experts, considering her current tumor status and medical history, anticipated potential adverse reactions during the treatment process and discussed handling protocols. Yun’s entire journey with CAR-T cell therapy was challenging. The medical team meticulously coordinated their efforts, successfully addressing adverse reactions after CAR-T treatment, ultimately averting potential risks.”
Despite lying in her hospital bed, Yun deeply appreciates the relentless dedication of her medical caregivers. “Sometimes, directors come to see me after 10 p.m., telling me they worked late but had to check on me; some arrive by my bedside as early as 7 a.m., concerned about my well-being. I continuously remind myself that I must strive, persist, and live up to the efforts made by doctors and my entire family,” said the 78-year-old Yun . “I made it through.”
#CARTCellSuccess #CancerWarrior #CompleteRemissionChronicles #FamilySupportMatters #CAR-TBreakthrough #InspiringHealthJourney #MedicalMiracles #NeverTooLateToFight #Cancerfight #Bloodcancer
#lymphoma
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79
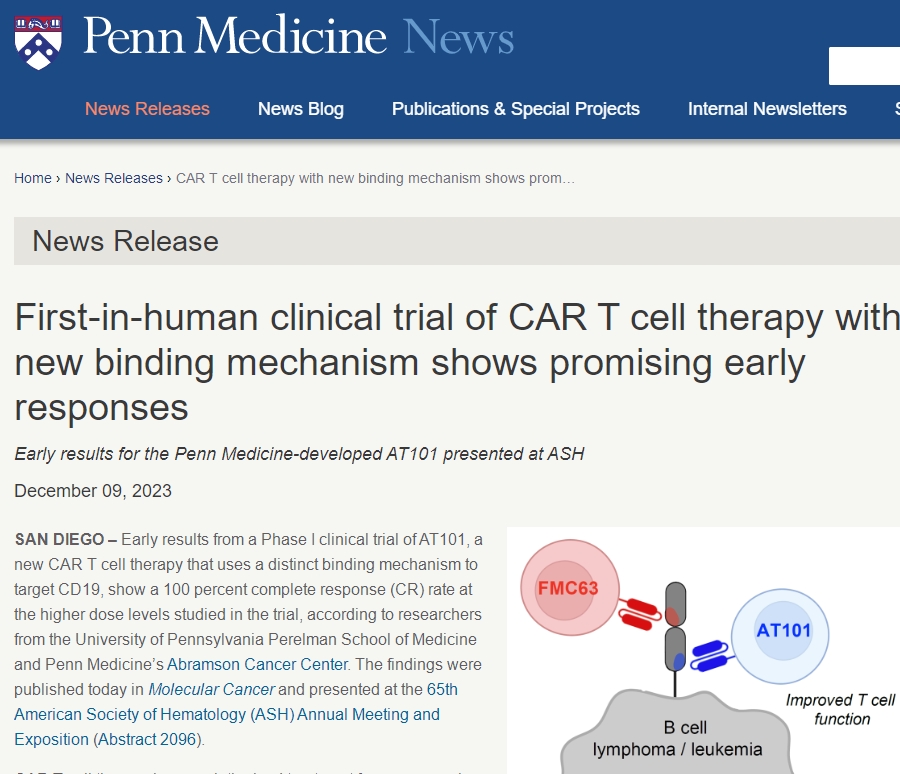
New Generation CAR-T Cell Therapy AT101: Phase I Clinical Trial Shows 100% Complete Remission Rate!
Researchers recently released exciting news: the Phase I clinical trial of their newly developed CAR-T cell therapy, AT101, demonstrated a 100% complete remission rate among patients receiving high-dose treatment. This groundbreaking achievement was published in the latest “Molecular Cancer” journal and gained significant attention at the 65th American Society of Hematology (ASH) Annual Meeting.
Despite recent FDA investigations into the safety of CAR-T cell therapy, it remains the most promising choice for blood cancer patients who have tried other treatment methods unsuccessfully. CAR-T cell therapy has fundamentally changed the treatment landscape for many blood cancer patients. While some patients show long-term responses to it, others do not.
The new CAR-T cell therapy, AT101, developed by researchers from the Perelman School of Medicine and the Abramson Cancer Center at the University of Pennsylvania, exhibited highly encouraging results due to its design targeting a new epitope of CD19 through a unique binding mechanism. Most currently approved CD19 CAR-T cell therapies target the same epitope, yet many patients eventually relapse. AT101, by targeting a different CD19 epitope, shows faster action rates and aims to reduce the failure rate of CAR-T cell therapy while improving clinical efficacy.
In this Phase I clinical trial, 14 patients with relapsed or refractory B-cell non-Hodgkin lymphoma (NHL) received treatment. Nine out of twelve patients achieved a complete remission status, indicating a total remission rate of 91.7%, with eight patients achieving complete remission. These patients did not experience cancer relapse, and the drug showed promising safety results.
While further research and larger-scale clinical trials are necessary, these early results instill considerable confidence. The success of AT101 holds promise for bringing new hope to blood cancer patients, especially those who previously did not receive effective CAR-T cell therapy. This study also aims to expand to a broader patient population, offering more treatment options and possibilities.
#CARTTherapy #BloodCancerTreatment #AT101Research #CancerRemission #MedicalBreakthrough #ASHAnnualMeeting #ClinicalTrialSuccess #UniversityResearch #CancerResearchUpdate #HopeForPatients #AT101 #BloodCancer #CancerTreatment
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

Recently, Professor Huang Huiqiang’s team at Sun Yat-sen University Affiliated Cancer Hospital achieved significant progress in a lymphoma patient who relapsed after five lines of chemotherapy. Utilizing CAR-T therapy, the patient has experienced complete remission for over two years, making them one of the patients in China with the longest survival post-CAR-T treatment.

Lymphoma, one of the common blood cancers, has traditionally been treated using a combination of chemotherapy, autologous hematopoietic stem cell transplantation, targeted therapies, and immune-modulating drugs. However, CAR-T cell therapy, an advanced technology in the field of oncology, has emerged as a precise, rapid, and highly effective new treatment method in recent years.
Diffuse large B-cell lymphoma is a prevalent aggressive lymphoma characterized by rapid progression, high mortality rates, and short survival periods. Despite significant therapeutic advancements in recent years, approximately 70% of patients achieve good outcomes and long-term remission through first-line treatments, but around 30% of patients have inadequate responses to treatment, facing difficult-to-cure relapses.

Professor Huang Huiqiang emphasized, “CAR-T cell therapy plays a crucial role in providing new treatment hopes for relapsed and refractory patients who cannot undergo transplantation or have previously undergone ineffective treatments.
It is reported that in the future, Sun Yat-sen University Cancer Center will continue to explore clinical innovative technologies in CAR-T cell therapy, aiming to bring new prospects for numerous patients.
#CARTBreakthrough #CancerResearch #SunYatSenCancerCenter #InnovativeTherapies #MedicalBreakthrough #LymphomaTreatment #PrecisionMedicine #HopeForPatients #CancerSurvivorship #AdvancedOncology #MedicalInnovation #ResearchProgress #ScienceNews #HealthcareAdvancements #ImmunotherapySuccess #Cancer