Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

The Chinese CAR-T therapy achieves a miraculous cure for advanced liver cancer, creating wonders in the field of cellular treatment for solid tumors.
The Chinese CAR-T therapy achieves a miraculous cure for advanced liver cancer, creating wonders in the field of cellular treatment for solid tumors.🌞🌞

🥰Surviving Against the Odds: A Chinese Doctor’s Journey with Liver Tumor🥰
Zou began his career in 1989 and has dedicated 30 years as an obstetrician-gynecologist, tirelessly working on the frontline of clinical care.
⭐️”Prof.Shi mentioned this immunotherapy, an antibody treatment,” said Dr. Zou’s wife.⭐️

Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

🥰Chinese CAR-T Therapy: A Beacon of Hope for Lymphoma Patients🥰
🥰Chinese CAR-T Therapy: A Beacon of Hope for Lymphoma Patients🥰
🌞In summary, CAR-T therapy presents a promising avenue for patients facing challenging lymphoma scenarios. The successful case of Mr. Li underscores the potential efficacy and safety of this innovative treatment at Peking Union Medical College Hospital.

Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79
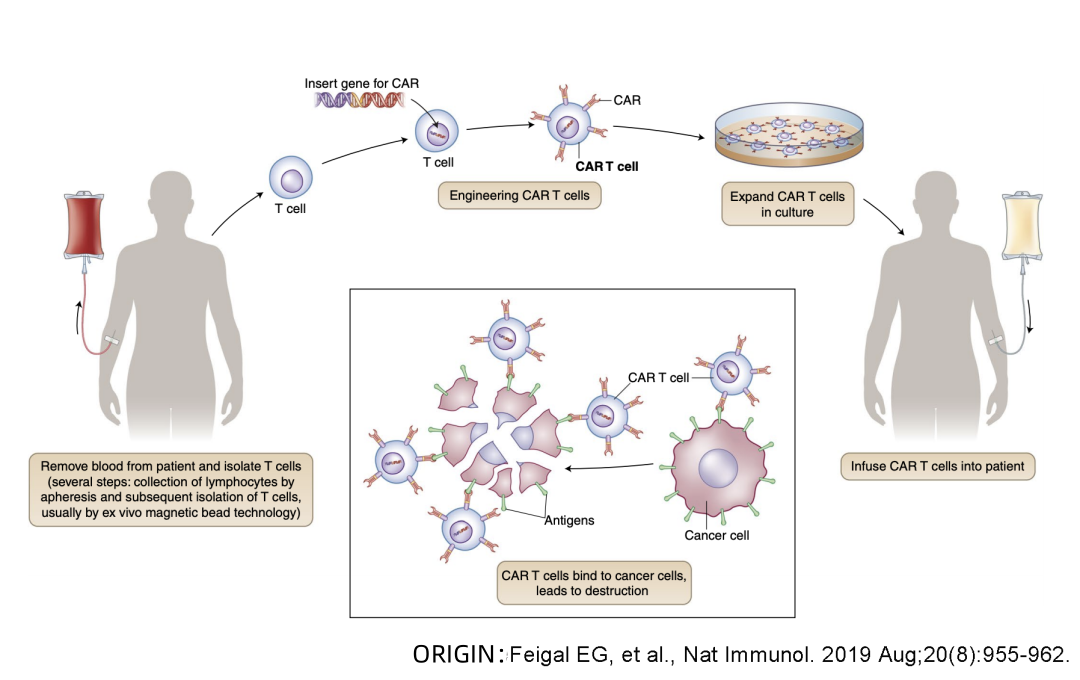
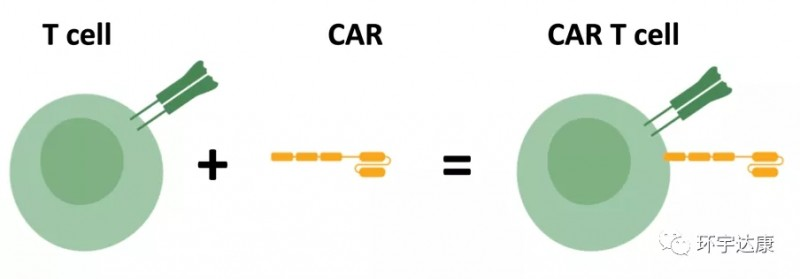
Beacon of Hope in Cancer Treatment – What is the treatment process for CAR-T cell therapy?
Beacon of Hope in Cancer Treatment
😊😊😊 What is the treatment process for CAR-T cell therapy?

Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

A Miraculous Journey: Israeli Artist Finds Cure for Multiple Myeloma in Hangzhou China






Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79
Breakthrough CAR-T Cell Therapy for R/R B-Cell ALL: A Game-Changer in Chinese Medical Innovation


Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

Clinical Breakthrough: Chinese CAR-T – Inaticabtagene Autoleucel Revolutionizing Hematologic Cancer Therapy


Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

Exploring Tumor Vitality: Chinese CAR-T Therapy Grants Patients Complete Remission


Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

Two Years of Complete Remission(Lymphoma), CAR-T therapy has given her a new lease on life.
“Two Years of Complete Remission, CAR-T therapy has given her a new lease on life. She Thought the Journey Was Over”

Yun is a 78-year-old patient who achieved a continuous complete remission for two and a half years despite relapsed/refractory diffuse large B-cell lymphoma. Diagnosed in 2019, after enduring six rounds of chemotherapy, she initially achieved complete remission as confirmed by her healthcare team. Unfortunately, the period of remission was short-lived as Yun soon experienced disease relapse, and subsequent second-line treatments failed to yield positive results.
“I vividly remember when Director Wang Li encouraged me, mentioning an advanced method called Car-T, but it was quite expensive. I was hesitant, but I discussed it with my son when I got home. When my son and daughter-in-law heard about it, they insisted on treatment. My son said, ‘Mom, you’re the only mother I have in this world. As long as you’re here, our home is complete. Money can be earned again, and if the treatment isn’t successful, at least we won’t have regrets.’ I was deeply moved. At my age, I’ve already shown strength through previous treatments. I believe that wherever my health takes me is where I belong. Everyone’s support gave me a reason to stay strong again.”
In 2021, CAR-T cell therapy was approved and launched in China, and Yun underwent this treatment at Ruijin Hospital.
Dr. Wang Li, Director of Hematology at Ruijin Hospital, explained, “CAR-T cell therapy provides new treatment options for a wide range of cancer patients.”
“Before planning Yun’s CAR-T cell therapy, lymphoma experts, considering her current tumor status and medical history, anticipated potential adverse reactions during the treatment process and discussed handling protocols. Yun’s entire journey with CAR-T cell therapy was challenging. The medical team meticulously coordinated their efforts, successfully addressing adverse reactions after CAR-T treatment, ultimately averting potential risks.”
Despite lying in her hospital bed, Yun deeply appreciates the relentless dedication of her medical caregivers. “Sometimes, directors come to see me after 10 p.m., telling me they worked late but had to check on me; some arrive by my bedside as early as 7 a.m., concerned about my well-being. I continuously remind myself that I must strive, persist, and live up to the efforts made by doctors and my entire family,” said the 78-year-old Yun . “I made it through.”
#CARTCellSuccess #CancerWarrior #CompleteRemissionChronicles #FamilySupportMatters #CAR-TBreakthrough #InspiringHealthJourney #MedicalMiracles #NeverTooLateToFight #Cancerfight #Bloodcancer
#lymphoma
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79
Breakthrough Advances in CAR-T Cell Therapy Combined with TKI for Malignant Hematologic System Tumors



Global First: Chinese CAR-T Therapy Simultaneously Cures Tumor and Lupus Erythematosus

Unveiling Hope: Chinese CAR-T Cell Therapy Illuminating New Paths in Liver Cancer Treatment!
2020, over 19.3 million people were diagnosed with cancer worldwide, leading to almost 10 million fatalities. In China alone, the number of new cancer patients reached a staggering 4.57 million, accounting for 23.7% globally. Liver cancer, among the most prevalent malignant tumors in China, witnessed 410,000 new cases and 380,000 deaths, making up 45.3% and 47.1% of the global total, respectively [1]. However, since the 21st century began, significant strides have been made in liver cancer treatments, particularly in medication and localized therapies. Surgical procedures are no longer the sole option for long-term survival among liver cancer patients.
Immunotherapy has emerged as one of the most promising techniques for treating liver cancer, especially with advancements in tumor molecular biology. In 2013, “Science” magazine categorized immunotherapy as the fourth major cancer treatment, following surgery, chemotherapy, and radiation therapy, with cell therapy becoming a focal point of basic and clinical research in recent years.

In May 2020, Professor Zhai Bo’s team from Shanghai Jiao Tong University School of Medicine’s Renji Hospital, in collaboration with Shanghai Sci-Tech Biotechnology’s team led by Li Zonghai, published groundbreaking preliminary clinical research data on CAR-T cell therapy targeting the GPC3 gene for hepatocellular carcinoma in the “Clinical Cancer Research” journal. This breakthrough study brought unprecedented hope for CAR-T cell therapy in liver cancer treatment.

Even more inspiring, their publication in the “Cancer Communications” journal showcased follow-up results of two late-stage liver cancer patients who achieved long-term tumor-free survival after receiving CAR-T cell combined with local therapy [2]. These findings shed new light on the treatment prospects for liver cancer patients.
However, despite the potential therapeutic effects of liver cancer CAR-T cell therapy, it faces challenges and obstacles. Liver cancer’s heterogeneity, tumor microenvironment, and the safety of cell therapy remain crucial issues to address.
Presently, revolutionary changes are underway in the treatment models and concepts for liver cancer. However, integrating CAR-T cell therapy into actual liver cancer treatment requires further scientific exploration and clinical research. Researchers emphasize that only through comprehensive utilization of CAR-T cells in conjunction with other treatment modalities can its therapeutic potential be fully realized.
Professor Zhai Bo’s team is currently conducting various fundamental and clinical studies aimed at exploring additional possibilities for CAR-T cell therapy in solid tumors. These studies include phase I clinical research on EpCAM CAR-T cell combined with ablative therapy for gastrointestinal tumors, phase II clinical research on Claudin18.2 CAR-T cell therapy for gastrointestinal tumors, studies on the mechanism and prevention of OTOT toxicity, among others. These endeavors will provide more experimental data and support for the application of CAR-T cells in the treatment of solid tumors.
In conclusion, liver cancer CAR-T cell therapy signifies a significant breakthrough in the field of liver cancer treatment, offering new hope for patients. Despite the challenges to overcome, the outlook for this therapy is promising and holds the potential to bring a blessing to more patients in the future.
[References]
Zhaibo, Lizonghai etc.
“Chimeric Antigen Receptor-Glypican-3 T-Cell Therapy for Advanced Hepatocellular Carcinoma: Results of Phase 1 Trials.” 《Clinical Cancer Research》, 2020.
“Combined local therapy and CAR-GPC3 T-cell therapy in advanced hepatocellular carcinoma: a proof-of-concept treatment strategy.” 《Cancer Communications》, 2023.
#HealthTech#CancerResearch #Immunotherapy #CARTcell #LiverCancer #MedicalBreakthrough #ClinicalTrials #ScienceNews #HealthcareInnovation #ResearchBreakthrough #MedicalScience #CancerTherapy #CancerAwareness #InnovativeMedicine #ImmunotherapyTreatment #ScienceUpdates #HealthcareTechnology #BiomedicalResearch #ClinicalInnovation #CancerTreatment #MedicalAdvancements #ImmunologyResearch #HealthcareIndustry #ProfessionalHealthcare
