Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79
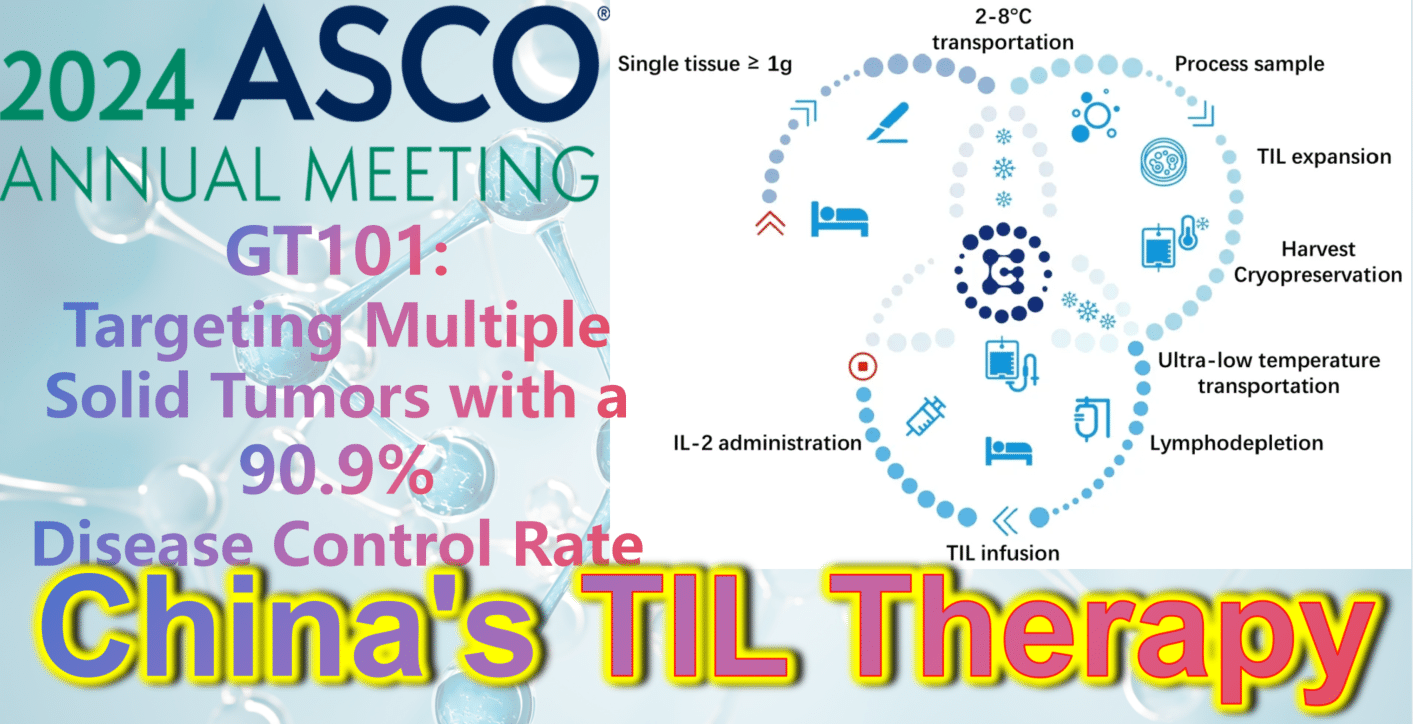
2024 ASCO China Highlights: China’s Indigenous TIL Therapy – GT101: Targeting Multiple Solid Tumors with a 90.9% Disease Control Rate
**2024 ASCO China Highlights: China’s Indigenous TIL Therapy Makes a Strong Debut, Targeting Small Cell Lung Cancer, Melanoma, Cervical Cancer, with a DCR Over 90%**
**GT101: Targeting Multiple Solid Tumors with a 90.9% Disease Control Rate**

TIL Therapy
GT101, independently developed by Gravel Biotech, is an autologous tumor-infiltrating lymphocyte (TIL) therapy. On April 22, 2022, its clinical trial implied consent was approved by the National Medical Products Administration (NMPA) (Acceptance No.: CXSL2200061). It is indicated for treating various solid tumors including non-small cell lung cancer, melanoma, and cervical cancer. Notably, GT101 is China’s first approved clinical TIL cell therapy and holds promise as the first cell therapy to conquer solid tumors!
Recently, at the 2024 ASCO Annual Meeting, results from the Phase 1 clinical trial of GT101 TIL therapy (NCT05430373) were announced. By November 10, 2023, a total of 14 patients with recurrent or metastatic solid tumors (including small cell lung cancer, melanoma, cervical cancer) had been enrolled, all with an ECOG performance status of 0 or 1 and having received a median of 2.6 prior lines of therapy. After enrollment, tumor tissue was obtained via appropriate surgical procedures for GT101 preparation, followed by approximately 30 days of TIL cell culture. Patients then underwent non-myeloablative lymphocyte depletion (cyclophosphamide + fludarabine), GT101 reinfusion therapy, and IL-2 (Interleukin-2) treatment.
1. **All enrolled patients (n=14):** The Objective Response Rate (ORR) was 35.7%. Among them, 28.6% (4 patients) achieved partial response (PR), 57.1% (8 patients) achieved stable disease (SD), and 7.1% (1 patient) achieved complete response (CR).
2. **In cervical cancer patients (n=11):** The Objective Response Rate (ORR) reached 45.5% (5/11). The Disease Control Rate (DCR) was as high as 90.9% (10/11), with 36.4% (4 patients) achieving partial response (PR) and 9.1% (1 patient) achieving complete response (CR). The median Progression-Free Survival (PFS) was 4.2 months. According to Kaplan-Meier statistics, the durations of complete response (CR) and progression-free survival (PFS) were 24 weeks and 36 weeks, respectively.
In conclusion, GT101 demonstrated promising clinical efficacy and manageable safety in combination with lymphocyte depletion and high-dose IL-2 treatment. Particularly in the treatment of cervical cancer, its objective response rate and duration of response are remarkable!
**How to Seek Help from TIL Therapy?**
The good news is that several TIL therapy clinical trials are currently recruiting in China, primarily targeting various solid tumors such as non-small cell lung cancer, melanoma, cholangiocarcinoma, esophageal squamous cell carcinoma, head and neck squamous cell carcinoma, breast cancer, ovarian cancer, cervical cancer, endometrial cancer, fallopian tube cancer, urothelial cancer, and renal cancer.
Patients seeking help from TIL therapy can submit their complete treatment history, recent pathology reports, imaging examination reports, and discharge summaries to Advanced Medicine in China
WhatsApp: +8613717959070
Email: doctor.huang@globecancer.com
#ASCO2024 #TILTherapy #GT101 #CancerResearch #SolidTumors #ClinicalTrials #LungCancer #Melanoma #CervicalCancer #Immunotherapy #ChinaBiotech #CancerTreatment
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

No Tumor Recurrence for Nearly 3 Years, China’s National Research #TIL Therapy #GC101 Targets Various Solid Tumors!
 No Tumor Recurrence for Nearly 3 Years, China’s National Research #TIL Therapy #GC101 Targets Various Solid Tumors!
No Tumor Recurrence for Nearly 3 Years, China’s National Research #TIL Therapy #GC101 Targets Various Solid Tumors!

TIL Therapy
 In recent years, immunotherapy has emerged as a pivotal direction in the future of cancer treatment. Tumor-infiltrating lymphocytes (#TILs) are hailed as the “natural enemies” of cancer cells. TIL therapy developed based on patients’ own tumor-infiltrating lymphocytes boasts advantages such as rich tumor-specific targets, excellent tumor homing capabilities, strong infiltration abilities, and a high safety profile.
In recent years, immunotherapy has emerged as a pivotal direction in the future of cancer treatment. Tumor-infiltrating lymphocytes (#TILs) are hailed as the “natural enemies” of cancer cells. TIL therapy developed based on patients’ own tumor-infiltrating lymphocytes boasts advantages such as rich tumor-specific targets, excellent tumor homing capabilities, strong infiltration abilities, and a high safety profile.
 On February 16, 2024, #Iovance Biotherapeutics announced the #FDA approval of Lifileucel (LN-144) for the treatment of advanced melanoma progressing after PD-1 antibody therapy, marketed as #AMTAGVI™. This marks the global first approval of a TIL therapy and the first approval worldwide for T-cell therapy for solid tumors, marking a significant milestone! #LN144
On February 16, 2024, #Iovance Biotherapeutics announced the #FDA approval of Lifileucel (LN-144) for the treatment of advanced melanoma progressing after PD-1 antibody therapy, marketed as #AMTAGVI™. This marks the global first approval of a TIL therapy and the first approval worldwide for T-cell therapy for solid tumors, marking a significant milestone! #LN144
 On the domestic front in China, several companies have announced good news regarding the clinical approval of TIL pipelines. Overall, the enthusiasm for TIL therapies is on the rise and increasingly intense. Today, let me introduce to you the first independently developed TIL cell therapy #GC101 from Juncell Therapeutics.
On the domestic front in China, several companies have announced good news regarding the clinical approval of TIL pipelines. Overall, the enthusiasm for TIL therapies is on the rise and increasingly intense. Today, let me introduce to you the first independently developed TIL cell therapy #GC101 from Juncell Therapeutics.
 With no tumor recurrence for nearly 3 years, GC01TIL is the first autologous natural tumor-infiltrating lymphocyte injection developed by Juncell Therapeutics, which officially obtained clinical trial implicit approval from the National Medical Products Administration (#NMPA) on April 24, 2022.
With no tumor recurrence for nearly 3 years, GC01TIL is the first autologous natural tumor-infiltrating lymphocyte injection developed by Juncell Therapeutics, which officially obtained clinical trial implicit approval from the National Medical Products Administration (#NMPA) on April 24, 2022.
 Distinguished from Iovance’s approved AMTAGVI™ (#Lifileucel), innovative GC101TIL therapy requires patients to be treated in ordinary wards without the need for high-intensity pre-conditioning before TIL cell infusion or any IL-2 dosage post-infusion. This simplified clinical protocol ensures effective TIL cell proliferation within patients, greatly avoiding the risks associated with AMTAGVI™’s black box warnings (such as treatment-related deaths, persistent severe cytopenia, severe infections, cardiopulmonary and renal function damage), substantially improving the safety, convenience, and accessibility of TIL therapy.
Distinguished from Iovance’s approved AMTAGVI™ (#Lifileucel), innovative GC101TIL therapy requires patients to be treated in ordinary wards without the need for high-intensity pre-conditioning before TIL cell infusion or any IL-2 dosage post-infusion. This simplified clinical protocol ensures effective TIL cell proliferation within patients, greatly avoiding the risks associated with AMTAGVI™’s black box warnings (such as treatment-related deaths, persistent severe cytopenia, severe infections, cardiopulmonary and renal function damage), substantially improving the safety, convenience, and accessibility of TIL therapy.
 Currently, according to the latest revelations from Juncell Therapeutics, the Phase I clinical trial of Juncell Therapeutics’ autologous natural TIL cell injection GC101 is underway in eight authoritative tertiary hospitals in China, achieving significant efficacy.
Currently, according to the latest revelations from Juncell Therapeutics, the Phase I clinical trial of Juncell Therapeutics’ autologous natural TIL cell injection GC101 is underway in eight authoritative tertiary hospitals in China, achieving significant efficacy.
 GC101, targeting various types of advanced solid tumors including malignant melanoma, cervical cancer, and lung cancer, has demonstrated an objective response rate of over 35%. Four patients have achieved complete tumor remission (#CR), with the longest no-recurrence survival period approaching 3 years, without experiencing any severe adverse reactions related to treatment, significantly improving the safety, applicability, and accessibility of #TILtherapy. Let us look forward to more surprises that GC101 will bring in the future!
GC101, targeting various types of advanced solid tumors including malignant melanoma, cervical cancer, and lung cancer, has demonstrated an objective response rate of over 35%. Four patients have achieved complete tumor remission (#CR), with the longest no-recurrence survival period approaching 3 years, without experiencing any severe adverse reactions related to treatment, significantly improving the safety, applicability, and accessibility of #TILtherapy. Let us look forward to more surprises that GC101 will bring in the future!
 Currently, #Juncell Therapeutics’ GC101 clinical trial for the treatment of advanced solid tumors is actively recruiting :
Currently, #Juncell Therapeutics’ GC101 clinical trial for the treatment of advanced solid tumors is actively recruiting :
FOR FREE
 Key Inclusion Criteria:
Key Inclusion Criteria:
1. Aged 18-75 years, regardless of gender;
2. Definitive diagnosis of malignant solid tumors (including but not limited to #melanoma, #lungcancer, #cervicalcancer, #esophagealsquamouscellcarcinoma, #headandnecksquamouscellcarcinoma, #endometrialcancer);
3. Standard treatment failure or lack of effective treatment options;
4. At least 2 lesions, with the physical condition supporting minimally invasive surgical sampling.
 This clinical trial is free of charge for participants. If you wish to participate in this clinical trial, you need to submit your treatment history, recent imaging and blood test reports, infectious disease reports, and discharge summaries to <@Advanced Medicine in China for preliminary assessment.
This clinical trial is free of charge for participants. If you wish to participate in this clinical trial, you need to submit your treatment history, recent imaging and blood test reports, infectious disease reports, and discharge summaries to <@Advanced Medicine in China for preliminary assessment.
 Email: doctor.huang@globecancer.com,
Email: doctor.huang@globecancer.com,
 WhatsApp: +8613717959070
WhatsApp: +8613717959070
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 78
Warning: Trying to access array offset on value of type bool in /www/wwwroot/www.medtourcn.com/wp-content/themes/medical-directory/framework/theme/medicaldirectory-image.php on line 79

Breakthrough! Chinese Novel CAR-T Therapy Kills Tumors and Prevents Relapse!
🌟✨ **Breakthrough! Chinese Novel CAR-T Therapy Kills Tumors and Prevents Relapse!** ✨🌟

CAR-T Therapy
🔬 On January 2, 2024, a groundbreaking clinical study from China was published in *Nature*. This study, employing engineering design, enables CAR-T cells to secrete Interleukin-10 (IL-10), thereby enhancing metabolism within the tumor microenvironment. The modified IL-10 CAR-T cells increase oxidative phosphorylation in a mitochondrial acetoacetate carrier-dependent manner, resulting in complete regression of solid tumors and metastatic cancers, including colon cancer, breast cancer, melanoma, and pancreatic cancer. This breakthrough research offers new hope for cancer patients.
🌱 **The Miracle of IL-10** 🌱
The secretion of IL-10 promotes the proliferation and effector functions of CAR T cells, leading not only to the regression of solid tumors but also inducing stem cell-like memory responses in lymphoid organs, providing enduring protection against tumor re-attack. Specifically, IL-10 HER2 CAR-T cells achieved complete regression of MC38-HER2 tumors in mice, with a cure rate of 90%. In the case of melanoma, IL-10 TRP-1 CAR-T cells achieved a clearance rate of 60%, with significant success in treating the orthotopic B16F10 melanoma model.
🦠 **A Weapon against Relapse** 🦠
In addition to complete regression of solid tumors, IL-10 CAR-T cells demonstrated the ability to prevent relapse in immunodeficient mice. Mice treated with IL-10 CD19 hCAR-T cells for Raji or PANC1-CD19 tumors exhibited complete tumor regression without relapse, indicating stronger anti-tumor capabilities of IL-10 CAR-T cells in xenograft models. Particularly noteworthy is the effective elimination of pancreatic ductal adenocarcinoma (PDAC) tumors by IL-10 CD19 hCAR-T cells, resulting in complete response in all treated mice.
💊 **A Revolutionary Treatment Approach** 💊
These findings suggest that IL-10-expressing CAR-T cells are an effective immunotherapy against various solid tumors, capable of achieving complete regression in multiple synthetic and xenograft tumor models. What’s more exciting is that preliminary results indicate the metabolism-enhanced IL-10 CD19 CAR-T cell therapy developed by Leman Biotech requires extremely low treatment doses, consistently achieving complete remission in numerous relapsed/refractory lymphoma or leukemia patients, paving the way for a new era in cancer treatment.
✨ **A Beacon of Hope** ✨
This breakthrough study brings hope to cancer patients and demonstrates the immense potential of CAR-T therapy in cancer treatment. Looking ahead, further advancements in this technology promise to provide more opportunities for recovery and survival to patients worldwide. Let’s anticipate more breakthroughs together and strive towards conquering cancer
#all #CARTtherapy #CancerTreatment #RRMM #IL10 #Tumor #Nature #MedicalBreakthrough #CARTCELL #coloncancer #breastcancer #melanoma #pancreatic 🌟🔬💊
